Count primary hepatocytes using the Cellometer Vision
Primary hepatocytes can be used for the in vitro evaluation of metabolism, drug-drug interaction, drug transporter activity, and toxicity of drug candidates. Hepatocytes are the
The Cellometer Vision (or Triangle Research Labs Gigacyte Hepatometer) can be used to successfully count primary hepatocytes. After successful primary hepatocyte cell isolation, 20 ul of sample, mixed with AO/PI fluorescent stains, can be placed into the Cellometer cell counting chamber for accurate and reproducible counts.
predominant cell in the liver, comprising about 60% of liver cells and 80% of liver mass. One of the main hepatocyte functions is the production of bile. Hepatocytes are also responsible for the regulation of metabolism, detoxification, and the manufacture of important circulating proteins, making them a critical component of studies involving liver disease, drug metabolism, and toxicology. Primary hepatocyte cell isolation remains a challenge for researchers.
Primary hepatocytes are very difficult to maintain in culture, displaying a rapid decline in viability and liver-specific functions. Optimization of primary hepatocyte culture conditions, including extracellular matrices (ECM), culture media / supplements, and co-culture with other cell types required for the normal cell-cell interactions displayed in vivo (micro-patterned co-cultures, MPCC) have enabled researchers to maintain hepatocyte viability and function for several weeks in culture. Recent studies have also proven cryopreservation (for up to 4 years) as a viable method of maintaining access to viable, functional primary hepatocytes.
Some laboratories produce their own primary hepatocytes. Primary hepatocyte cell lines are are also commercially available from sources such as Zen-Bio (Research Triangle Park, NC), Gigacyte (Branford, CT), or Triangle Research Labs (Research Triangle Park, NC).
The Cellometer® Vision Cell Analyzer (or Cellometer Gigacyte Hepatometer) incorporates image based cell counting and fluorescence detection in a compact and easy-to-use instrument. With dual fluorescence detection capabilities, Cellometer Vision is an ideal solution for many complex cell population characterization assays, such as reliable counting and viability determination of primary hepatocytes.
Reliable concentration and viability determination of primary hepatocytes is critical for accurate analysis of compound toxicity in vitro. Due to hepatocytes’ variable morphology, fragile nature and tendency to clump, traditional manual counting methods are time-consuming and the subjectivity from operator-to-operator can cause inconsistent results.
Nexcelom’s new method incorporates a ready-to-use fluorescent dual staining solution that stains live cells with acridine orange, and dead cells with propidium iodide. Researchers then load 20μL of labeled sample into a disposable counting chamber for analysis. Because the counting chamber is disposable, no washing is required between samples and the risk of cross contamination is eliminated.
Fluorescent images of the stained cells are captured and analyzed using proprietary algorithms. Cellometer Vision’s robust operating software accurately analyzes cell images to generate live cell count, concentration and viability percentage. Total analysis time is typically less than 60 seconds. Cell images and all analysis data, including cell size distribution histograms, can be instantly saved for documentation. Data can also be easily exported to Microsoft Excel spreadsheets for further analysis.
Method
Treat cell sample with Nexcelom’s Fluorescence Dual Staining Solution:
1. Take 20μl of freshly isolated hepatocyte sample or freeze-thaw cryopreserved cell sample in a small microtube.
2. Apply 20µl of ready-to-use dual staining solution (acridine orange/propidium iodide cocktail).
3. Gently mix. Sample is ready to count.
Running Assay:
1. Load 20µl of labeled sample into the Cellometer disposable counting chamber.
2. Insert chamber into Cellometer Vision.
3. Select assay from drop-down menu and enter Sample ID
4. Preview cell images and click ‘Count’ to begin analyzing sample.
5. Review images and counting results.
6. Save or Export images and/or report data.
Results
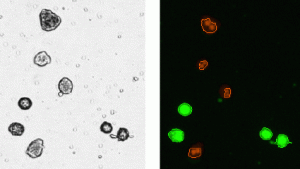
AO stained live hepatocytes are clearly visible in the fluorescent image obtained from Filter Set 101. The software indicates counted cells with a green circle (enlarged to show detail) while ignoring cellular debris. The software can also recognize and discretely count clumpy cells. PI stained dead cells are visible in the image obtained from Filter Set 202. The software then accurately calculates total cell count, concentration and viability (below). By using this method, live and dead cells are clearly distinguished and automatically counted for improved accuracy. By combining a ready to use staining solution and imaging based system results can be obtained much easier and faster compared to other methods.
Download the application note.

Leave a Reply
You must be logged in to post a comment.